 Reverse Osmosis (RO) was developed in the late 1950’s under U.S. Government funding, as a method of desalinating seawater. Reverse osmosis (RO) is a separation process that uses pressure to force a solvent (water) through a membrane, which retains the solute (contaminant) on one side and allows the pure solvent (water) to pass to the other side. More formally, it is the process of forcing a solvent from a region of high solute concentration through a membrane to a region of low solute concentrate by applying pressure in excess of osmotic pressure. This process is the reverse of the normal osmosis process, which is the natural movement of solvent from an area of low solute concentration, through a membrane, to an area of high solute concentration when no external pressure is applied. The membrane here is semi-permeable, meaning it allows the passage of solvent (water) but not of solute (contaminant).
Reverse Osmosis (RO) was developed in the late 1950’s under U.S. Government funding, as a method of desalinating seawater. Reverse osmosis (RO) is a separation process that uses pressure to force a solvent (water) through a membrane, which retains the solute (contaminant) on one side and allows the pure solvent (water) to pass to the other side. More formally, it is the process of forcing a solvent from a region of high solute concentration through a membrane to a region of low solute concentrate by applying pressure in excess of osmotic pressure. This process is the reverse of the normal osmosis process, which is the natural movement of solvent from an area of low solute concentration, through a membrane, to an area of high solute concentration when no external pressure is applied. The membrane here is semi-permeable, meaning it allows the passage of solvent (water) but not of solute (contaminant).

Water & Waste Management
 Sewage treatment is the process of removing contaminants from wastewater and household sewage, both runoff (effluents), domestic, commercial and institutional. It includes physical, chemical, and biological processes to remove physical, chemical and biological contaminants. Its objective is to produce an environmentally safe fluid waste stream (or treated effluent) and a solid waste (or treated sludge) suitable for disposal or reuse (usually as farm fertilizer). Using advanced technology it is now possible to re-use sewage effluent for drinking water
Sewage treatment is the process of removing contaminants from wastewater and household sewage, both runoff (effluents), domestic, commercial and institutional. It includes physical, chemical, and biological processes to remove physical, chemical and biological contaminants. Its objective is to produce an environmentally safe fluid waste stream (or treated effluent) and a solid waste (or treated sludge) suitable for disposal or reuse (usually as farm fertilizer). Using advanced technology it is now possible to re-use sewage effluent for drinking water
 Greywater, is generally accepted as being wastewater generated from wash hand basins, showers and baths, which can be recycled on-site for uses such as WC flushing, landscape irrigation and constructed wetlands. Greywater often excludes discharge from laundry, dishwashers and kitchen sinks due to the high nutrient levels.
Greywater, is generally accepted as being wastewater generated from wash hand basins, showers and baths, which can be recycled on-site for uses such as WC flushing, landscape irrigation and constructed wetlands. Greywater often excludes discharge from laundry, dishwashers and kitchen sinks due to the high nutrient levels.
 Ultrafiltration (UF) is a variety of membrane filtration in which hydrostatic pressure forces a liquid against a semipermeable membrane. Suspended solids and solutes of high molecular weight are retained, while water and low molecular weight solutes pass through the membrane. This separation process is used in industry and research for purifying and concentrating macromolecular (103 - 106 Da) solutions, especially protein solutions. Ultrafiltration is not fundamentally different from microfiltration, Nano filtration or gas separation, except in terms of the size of the molecules it retains. Ultrafiltration is applied in cross-flow or dead-end mode and separation in ultrafiltration undergoes concentration polarization.
Ultrafiltration (UF) is a variety of membrane filtration in which hydrostatic pressure forces a liquid against a semipermeable membrane. Suspended solids and solutes of high molecular weight are retained, while water and low molecular weight solutes pass through the membrane. This separation process is used in industry and research for purifying and concentrating macromolecular (103 - 106 Da) solutions, especially protein solutions. Ultrafiltration is not fundamentally different from microfiltration, Nano filtration or gas separation, except in terms of the size of the molecules it retains. Ultrafiltration is applied in cross-flow or dead-end mode and separation in ultrafiltration undergoes concentration polarization.
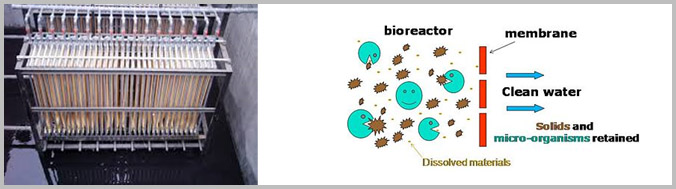
Membrane bioreactor (MBR) is the combination of a membrane process like microfiltration or ultrafiltration with a suspended growth bioreactor, and is now widely used for municipal and industrial wastewater treatment

An oil-water separator is a device designed to separate gross amounts of oil and suspended solids from the wastewater effluents of oil refineries, petrochemical plants, chemical plants, natural gas processing plants and other industrial sources.

Gas flow is passed through the carbon adsorption system including one prefilter (demister) and one carbon scrubber, by a fan located at the inlet of the system. The clean air is discharged through the stack installed on top of scrubber. Water washable activated carbon is used in this system to remove 99% of H2S gas.
Carbon Adsorption Technique
H2S removal is effectively handled until about 25% of the carbon pores are used. This is the breakthrough point where the carbon must be replaced. The carbon vessel is protected against static charge build up by grounding the conductive strip in the vessel. A differential pressure gauge is utilized to indicate the differential pressure in the scrubber.

Water disinfection means the removal, deactivation or killing of pathogenic microorganisms. Microorganisms are destroyed or deactivated, resulting in termination of growth and reproduction. When microorganisms are not removed from drinking water, drinking water usage will cause people to fall ill.

Ozone is "active oxygen", nature's special element and a beautiful gift to mankind. Ozone is
a triatomic allotrope of oxygen formed by recombination of oxygen atoms. It is a colourless
gas with characteristic pungent odor and can easily be detected at low level of 0.02ppm.
It is a very strong oxidant and is over 3000 times more powerful disinfectant than chlorine.
Ozone is a one-stop solution to most of the water treatment related problems such as bacteria,
virus, algae, fungi, colour, odor, COD and BOD. It disinfects, oxidizes, deodorizes and
decolorizes. Ozone is generated by an electrical discharge through dry air or pure oxygen
and is generated onsite because it decomposes to elemental oxygen in a short amount of time.

